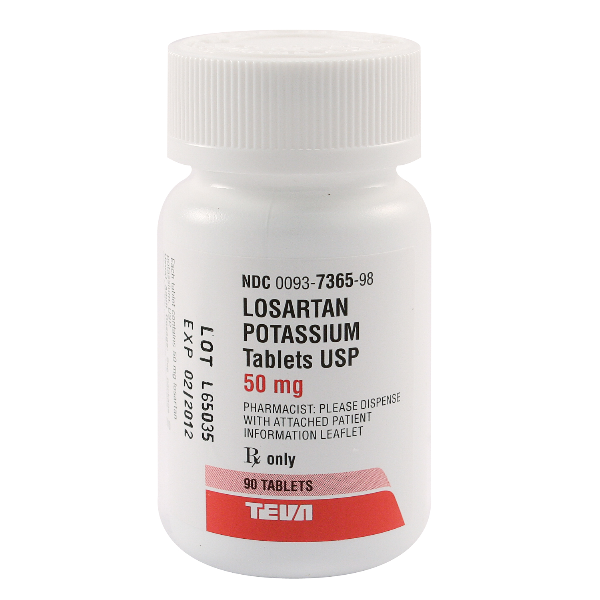
Teva Pharmaceuticals USA, Inc. Expands Voluntary Nationwide Recall of Losartan Potassium to 50 mg and 100 mg Tablets USP, Sold Exclusively to Golden State Medical Supply, Inc. | FDA

L'Aifa ritira due lotti di Losartan, farmaco contro l'ipertensione: “Presenza di impurezze” - La Stampa

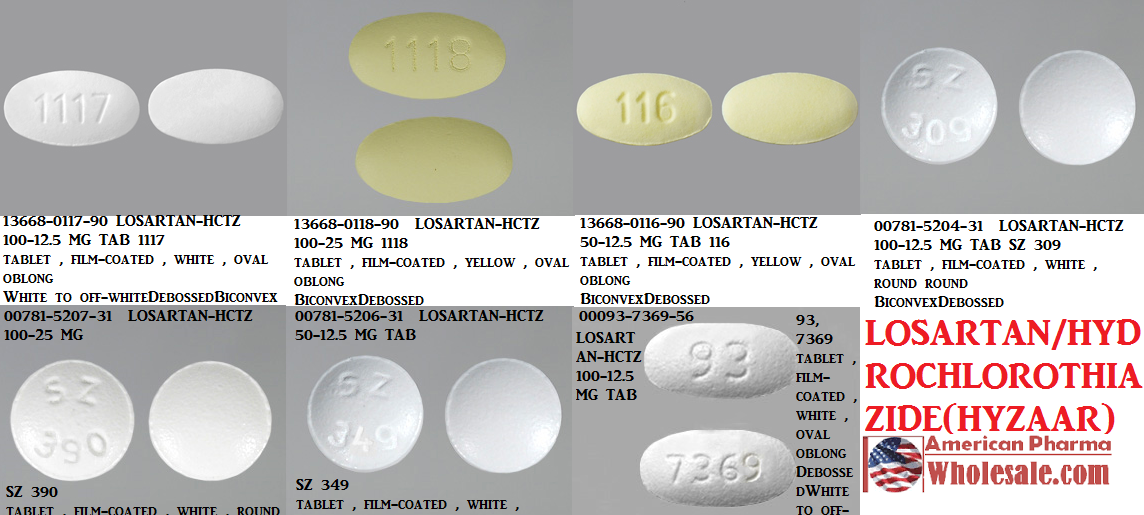
oval yellow 93 7367 Images - Losartan Potassium and Hydrochlorothiazide - losartan potassium and hydrochlorothiazide - NDC 0093-7369-98